 Reveal Awarded $3M in NIH Grants for Novel MRI Contrast Agent
Reveal Awarded $3M in NIH Grants for Novel MRI Contrast Agent
Reveal wins National Cancer Institute support for first in human clinical trials
Cambridge MA, 28 September 2021 - Reveal Pharmaceuticals, an innovative company developing the first general purpose gadolinium-free MRI contrast agent, is pleased to announce two substantial SBIR grants from the National Cancer Institute of the National Institutes of Health. One award, a Fast Track Phase II SBIR grant of $1M, is the culmination of an SBIR grant totaling $2.3M: this grant advances development of RVP-001 and extends its application to liver imaging. The other award, a Direct to Phase II SBIR grant of up to $2M, will support first in human clinical trials for RVP‑001.
Each year over 40 million contrast-enhanced MRIs are performed, providing vital insight into many serious health conditions. Current MRI contrast agents
all cause accumulation of the heavy metal gadolinium in the brain and body of every patient and are also driving rising levels of anthropogenic gadolinium
in earth’s surface waters. Reveal’s first in class gadolinium-free MRI contrast agent, RVP‑001, is designed to be a direct replacement for current MRI contrast agents,
promising to benefit tens of millions of patients.
“We appreciate the National Cancer Institute’s tremendous support for Reveal’s important work. NCI’s sponsorship underscores the significance of RVP‑001
and its potential to positively impact human health” said Vera Hoffman, CEO of Reveal and Principal Investigator on both grants.
“This funding will help advance our commitment to patients by providing a novel gadolinium free agent which avoids the risks and potential late effects of
gadolinium inherent to all currently approved MRI contrast agents” said Srinivasan Mukundan, Reveal’s Medical Director. “This is very important to patients
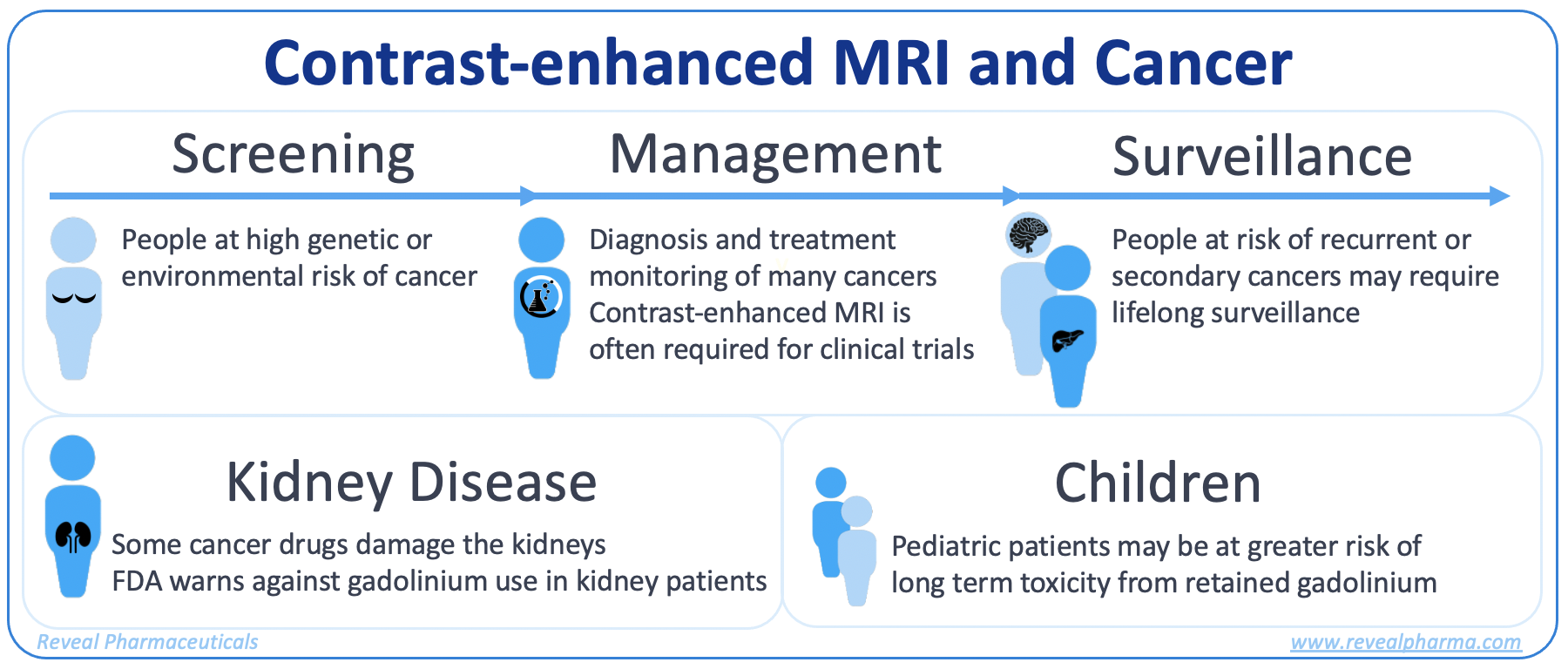
in vulnerable groups such as people who need repeated MRI scans, people with kidney disease, and pediatric patients. In particular, people facing cancer may
require dozens of contrast-enhanced MRI scans for screening, surveillance, to guide treatment, and as part of many clinical trials.”
These two National Cancer Institute (NCI) SBIR grants complement previous work funded by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) and the National Heart Lung,
and Blood Institute (NHLBI), reflecting the broad importance of Reveal’s novel contrast agent for many serious health conditions. Reveal has also won support and recognition
from the Massachusetts Life Sciences Center, MassVentures, MassChallenge, Endless Frontier Labs, and the Harvard Business School Alumni New Venture Competition.
Reveal is transforming medical insight, improving human health, and positively impacting the environment. Over 40M contrast-enhanced magnetic resonance imaging (MRI) scans are performed each year.
Current MRI contrast agents all cause accumulation of the heavy metal gadolinium in the brain and body of every patient, and drive rising levels of gadolinium in surface waters.
Reveal’s patented contrast agents use manganese, which is essential for life. Reveal’s IND-ready lead product RVP-001 promises to directly replace current MRI contrast agents, benefiting patients and the planet.
NCI leads the National Cancer Program and NIH’s efforts to dramatically reduce the prevalence of cancer and improve the lives of cancer patients and their families. For more information, please visit the NCI website at cancer.gov.
NIH, the nation's medical research agency, is the primary federal agency conducting and supporting basic, clinical, and translational medical research; investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit nih.gov.
Disclaimer
The grants reported in this press release are supported by the National Cancer Institute of the National Institutes of Health under award numbers R44CA239935 and R44CA268392. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.

